
Introduction
GS1 serialization appears in compliance documents and vendor pitches constantly — yet it's rarely explained at a practical, operational level. For supply chain managers, compliance officers, and brand operations teams across FMCG, food, and pharma, that gap has real consequences:
- Platform listing rejections due to incorrect identifier formats
- Shipment refusals from downstream distributors
- Failed regulatory verification requests during audits
GS1 serialization is the process of assigning a globally unique identifier to each individual product unit, enabling item-level tracking across the supply chain. It underpins traceability, anti-counterfeiting enforcement, and regulatory compliance — from India's FSSAI traceability requirements and CDSCO drug serialization mandates to DSCSA in the US and the Falsified Medicines Directive in the EU.
This guide covers what GS1 serialization is, how it works end-to-end, and where it applies in your operations — moving beyond compliance checklists to operational implementation at each stage of your supply chain.
TL;DR
- GS1 serialization assigns a unique identifier (GTIN + serial number + batch/lot + expiry date) to every individual product unit, not just the product type
- Required under DSCSA (USA) and the Falsified Medicines Directive (EU), and now standard in global FMCG and food supply chains
- Compliance requires printing a GS1 DataMatrix or GS1-128 barcode at manufacture, logging events via EPCIS, and sharing custody data across the chain
- Serialization tracks individual units; aggregation links them to cases and pallets via SSCC — these are separate, sequential processes
- Most compliance failures trace to data inconsistencies, outdated EPCIS versions, or poor system integration, not flaws in the standard itself
What Are GS1 Serialization Standards?
GS1 serialization standards are globally maintained rules that specify how products are uniquely identified, barcoded, and tracked at the individual unit level. GS1, the international not-for-profit standards organization, oversees these standards through 120 local member organizations spanning 150 countries. The four core data elements are the Global Trade Item Number (GTIN), a unique serial number, a batch/lot number, and an expiration date.
The Four Key GS1 Components for Serialization
These components form an integrated system — each element plays a distinct role in tracking a product from manufacture to end consumer:
| Component | Purpose | Application Identifier |
|---|---|---|
| GTIN (Global Trade Item Number) | Identifies the specific product, embedding the National Drug Code (NDC) in US pharma | AI (01) |
| Serial Number | A unique alphanumeric code assigned to an individual instance of a product for its lifetime | AI (21) |
| Batch/Lot Number | Associates an item with manufacturing traceability data | AI (10) |
| Expiration Date | Indicates the shelf life or use-by date of the product | AI (17) |

For unit-level serialization—prescription packages or the lowest saleable unit—the GS1 DataMatrix is the standard 2D barcode. For logistics units, cases, and outer packaging, the GS1-128 linear barcode is used.
EPCIS: The Data Exchange Backbone
Electronic Product Code Information Services (EPCIS) enables trading partners to capture and share interoperable information about the "what, when, where, why, and how" of products as they move through the supply chain. EPCIS 2.0, ratified in March 2023, introduced developer-friendly JSON/JSON-LD syntax and GS1 Digital Link URIs, moving away from legacy XML and EPC URNs.
Serialization vs. Aggregation: Distinct Processes
Serialization assigns a unique identity to every individual unit (for example, one pill bottle). Aggregation links serialized units into higher packaging levels—cases, cartons, pallets—using the Serial Shipping Container Code (SSCC). The SSCC is an 18-digit GS1 identifier assigned to logistics units, encoded in a GS1-128 barcode with Application Identifier (00).
Aggregation is a complementary process built on top of serialization. A GTIN combined with a serial number identifies an individual unit — it does not replace the SSCC, which is the identifier for a logistics unit.
Why GS1 Serialization Matters in the Supply Chain
Regulatory Mandates and Compliance Deadlines
In India, GS1 barcodes are now a practical requirement for brands selling through Quick Commerce platforms, modern trade, and organized retail. FSSAI mandates accurate product identification on packaged foods, and the Legal Metrology (Packaged Commodities) Rules require compliant labeling with verifiable product data — both of which align directly with GS1 standards. GS1 India, operating under the Department for Promotion of Industry and Internal Trade (DPIIT), is the country's official GS1 member organization and the authoritative source for GTIN allocation.
Globally, the regulatory picture is equally demanding. In the US, the Drug Supply Chain Security Act (DSCSA) requires item-level serialization for all prescription drugs, with full enforcement active since November 2024. In the EU, Directive 2011/62/EU mandates GS1 DataMatrix codes on prescription packaging.
Recent enforcement actions in multiple markets show the consequences are real: distributors have received warning letters for failing to verify authorized trading partners or maintain product investigation records.
Precision Traceability and Recall Efficiency
Each serialized unit carries a unique digital fingerprint verifiable at any point in the supply chain — from manufacturer to distributor to retailer or dark store. When a recall is triggered, only the affected units are flagged and removed.
Without serialization, recalls default to lot-wide removal: operationally expensive and far broader than necessary. EPCIS-based traceability lets supply chain partners isolate the exact affected batch, act quickly, and document recall progress in real time.
Anti-Counterfeiting Protection
Serialized products with unique identifiers verified against a manufacturer's database are impossible to replicate without triggering detection. The scale of the problem makes this protection critical:
- The WHO estimates 1 in 10 medicines in low- and middle-income countries are substandard or falsified, costing an estimated $30.5 billion annually
- The Pharmaceutical Security Institute (PSI) recorded 6,424 pharmaceutical crime incidents in 2024 alone
- Counterfeit FMCG products — including spices, packaged foods, and personal care — follow the same distribution channels as genuine goods, making serialization the only reliable detection layer
Operational Efficiency Gains
For FMCG brands operating across Quick Commerce platforms, modern trade, and regional distributors, GS1 standardization directly reduces operational drag. Standardized identifiers enable:
- Automated inventory replenishment triggered by scan events
- Accurate expiry date tracking at SKU level
- Reduced shrinkage across warehousing and last-mile distribution
- Consistent product data across Blinkit, Zepto, Swiggy Instamart, and JioMart catalogs
These gains apply across dairy, masalas, staples, snacks, and personal care — any category where speed of replenishment and shelf availability directly affect platform rankings.
What Goes Wrong Without Serialization
- Inability to fulfill DSCSA verification requests from trading partners
- Shipment rejection by downstream distributors who cannot confirm product authenticity
- Inability to achieve end-to-end traceability during contamination events or recalls
- Platform listing rejections on Quick Commerce networks that mandate GS1-compliant barcodes
How GS1 Serialization Works: The End-to-End Process
GS1 serialization follows a defined sequence: identifiers are generated before production, encoded during manufacturing, and tracked at every custody transfer point using EPCIS. Each stage builds on the last, creating an unbroken chain of traceability from factory floor to end destination.
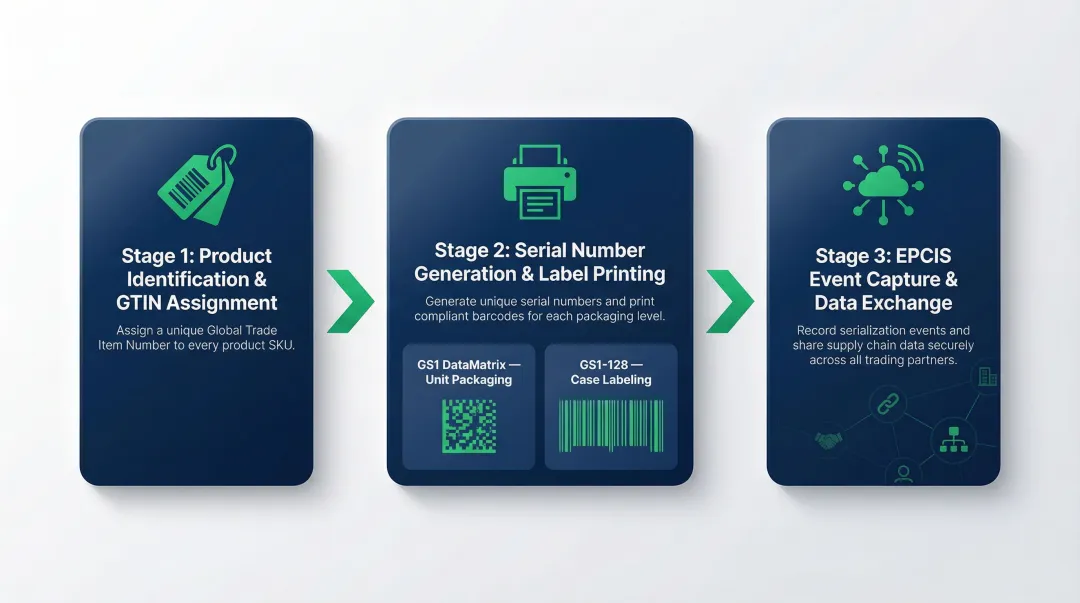
Step 1: Product Identification and GTIN Assignment
Every product variant requiring serialization must first have a GTIN—a globally unique product identifier obtained through a GS1 company prefix. GTIN assignment happens at the product level (not the unit level): the same GTIN applies to all units of a given SKU, while the serial number differentiates individual units.
To get a GTIN, a brand registers with a local GS1 Member Organization and receives a company prefix — the building block for all GS1 identification keys. GTINs can be 8, 12, 13, or 14 digits long, but are always stored and encoded in a 14-digit format (using leading zeros where needed).
Step 2: Serial Number Generation and Label Printing
Unique serial numbers are generated—either by the manufacturer's serialization system or through a Level 4/5 software platform—and combined with the GTIN, batch/lot number, and expiry date to form a complete serialized identifier.
This composite data is encoded into:
- GS1 DataMatrix (2D barcode) for unit-level packaging
- GS1-128 (1D barcode) for cases and outer packaging
Print quality is critical. GS1 standards use ISO/IEC 15415 for measuring and grading two-dimensional symbols like the GS1 DataMatrix, and ISO/IEC 15416 for linear symbols like the GS1-128. Unreadable barcodes create downstream compliance failures.
Step 3: EPCIS Event Capture and Data Exchange
Once products are serialized and packed, supply chain events (commissioning, shipping, receiving, decommissioning) are captured and reported using EPCIS. EPCIS records the "what, when, where, and why" of each event, creating an auditable chain of custody.
EPCIS captures four event types:
| Event Type | Action | Supply Chain Context |
|---|---|---|
| ObjectEvent | ADD | Marks commissioning when a serial number is applied |
| AggregationEvent | ADD | Children are aggregated to a parent (packing items into a case) |
| ObjectEvent / AggregationEvent | OBSERVE | Objects are observed without creation or destruction (shipping/receiving) |
| ObjectEvent | DELETE | Marks decommissioning or destruction |
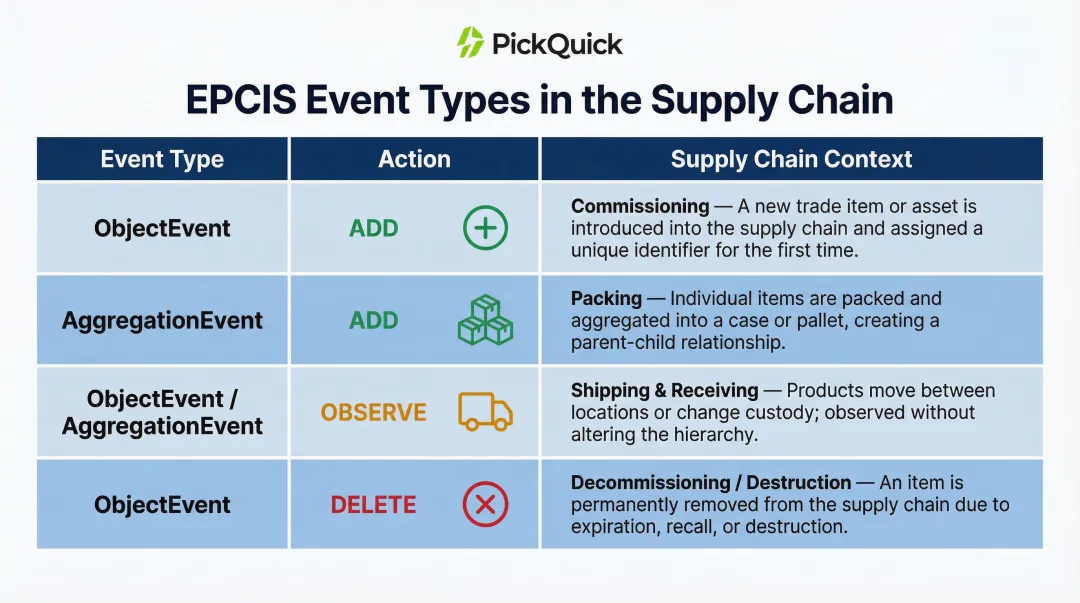
EPCIS data must be exchanged between all trading partners: manufacturers, 3PLs, distributors, and retailers. The version in use (for example, 1.2 vs. 2.0) must match across all parties to ensure interoperability.
Key Factors and Where GS1 Serialization Is Applied
Industries and Product Categories
GS1 serialization is applied across:
- Pharmaceuticals (most regulated—DSCSA and FMD mandates)
- Medical devices (UDI-mandated)
- FMCG and packaged foods (for supply chain visibility and retailer compliance)
- Consumer goods entering modern trade or Quick Commerce platforms
Brands selling across dark store networks—like those operating on Blinkit, Zepto, and Swiggy Instamart—need GS1-compliant product identification to maintain accurate inventory records across thousands of pincodes. Indian e-commerce platforms now strictly mandate GS1-standard barcodes (EAN/UPC/GTIN) for all product listings. For brands working with a Quick Commerce operator, GS1 readiness is typically handled as a prerequisite before inventory reaches any platform.
Key Implementation Factors
Several variables determine how a serialization program is designed and executed:
- Regulatory jurisdiction: DSCSA, FMD, and local mandates each carry different data requirements
- IT system readiness: Serialization requires integration across ERP, MES, WMS, and serialization software
- Production scale: High-volume lines need automated barcode printing and inline verification
- Trading partner readiness: If downstream partners cannot receive or validate EPCIS data, the system breaks at handoff
Serialization is a Recurring Process
Serialization is not a one-time event. It is a recurring, production-integrated process where every new batch requires:
- Serial number generation
- Label printing and verification
- Event capture and EPCIS reporting
Any change in product, packaging format, or market destination typically triggers a re-evaluation of the serialization setup.
Common Issues and Misconceptions in GS1 Serialization
Misconception: A Barcode Equals Full Compliance
The most common misconception is that printing a GS1 DataMatrix barcode on packaging equals full serialization compliance. True compliance requires the entire data chain—accurate serial number management, EPCIS event capture, and verified data exchange with trading partners. A barcode that is printed but not registered or communicated is operationally useless.
Frequent Operational Failures
Three failure types appear consistently across serialization rollouts:
- Data mismatches between the barcode and the manufacturer's database cause verification failures downstream
- Incorrect or outdated EPCIS versions that downstream partners cannot process create interoperability gaps
- Poor ERP integration between serialization software and ERP systems results in duplicate or missing serial records
Industry pilots reveal that "aggregation errors"—such as case-to-pallet mismatches where physical products do not match the transmitted EPCIS data—are a leading cause of supply chain disruption. These mismatches force downstream partners to quarantine products because they have no record of receiving the transaction data.

Aggregation Confusion
The case-to-pallet mismatch problem above often traces back to a gap in understanding aggregation. Many teams assume that completing unit-level serialization means aggregation is also handled. It is not. Aggregation—linking serialized units to cases and pallets via SSCC—is a separate process that requires its own configuration and execution. Skipping it creates problems during warehouse receipt and order verification workflows.
Conclusion
GS1 serialization is a globally standardized, multi-component process—not just a barcode format—that gives every product unit a unique, verifiable identity across the supply chain. Getting it right means treating four components as a single integrated system:
- GTIN assignment — unique product-level identification registered with GS1
- Serial number management — unit-level uniqueness enforced at the production stage
- Barcode encoding — correct symbology (GS1-128, DataMatrix, QR) for the channel
- EPCIS data exchange — event-level traceability shared across supply chain partners
Brands that treat GS1 compliance as a checklist exercise — barcode present, box ticked — tend to discover gaps only when a platform rejects a shipment or a recall surfaces missing traceability data. Audit your implementation against the full GS1 framework, not just barcode presence. That gap between surface compliance and operational readiness is where most serialization failures happen.
For brands scaling across Quick Commerce networks, GS1 readiness is no longer optional—it is the operational foundation for listing approval, replenishment accuracy, and expansion across dark stores.
Frequently Asked Questions
What is GS1 serialization and how does it differ from aggregation in the pharmaceutical industry?
GS1 serialization assigns a unique identifier (GTIN + serial number + batch + expiry) to each individual unit, while aggregation links those units to higher packaging levels (cases, pallets) using the SSCC. Both processes are required for full DSCSA compliance but serve different functions.
What FDA guidance applies to GS1 serialization standards?
The Drug Supply Chain Security Act (DSCSA) is the primary FDA regulatory driver for pharmaceutical serialization, while the UDI rule covers medical devices. GS1 US's implementation guidelines—including EPCIS 1.2 and the GS1 US Implementation Guideline for DSCSA—operationalize compliance for both.
What are GS1 serialization standards?
They are the internationally maintained technical rules governing how unique product identifiers are structured, encoded in barcodes (GS1 DataMatrix or GS1-128), and communicated via EPCIS to enable item-level traceability across the global supply chain.
What are the GS1 supply chain standards?
The core GS1 supply chain standards include GTIN (product identification), GLN (location identification), SSCC (logistics unit identification), GS1 barcodes (data carriers), and EPCIS (event data sharing). Together, these form a common language for supply chain visibility.
What is the GS1 SSCC standard?
The Serial Shipping Container Code (SSCC) is an 18-digit GS1 identifier assigned to logistics units such as pallets or cases. It is encoded in a GS1-128 barcode with Application Identifier (00) and used to track shipments through warehousing and distribution.
What are the three GS1 EDI standards?
The three primary GS1 EDI standards are GS1 XML, EANCOM (the GS1 subset of UN/EDIFACT), and the GS1 implementation of ASC X12 (used predominantly in North America). These govern how structured business documents—orders, invoices, shipment notices—are exchanged electronically between trading partners.


